Manufacture of Nitroguanidine
Nitroguanidine is manufactured in reactors by dehydrating guanidine salts in presence of sulfuric acid as a reaction agent and solvent.
Further processes are: Precipitation of the nitroguanidine by mixing with water in a precipitation apparatus which has a large influence on the quality and crystal size.
Separation and neutralization of the crystals on a rotating filter. Dewatering in the centrifuge and dryer.
Finally grinding.
The diluted sulphuric acid is concentrated in a multi-stage plant to the required acid strength and again used in the reactor.
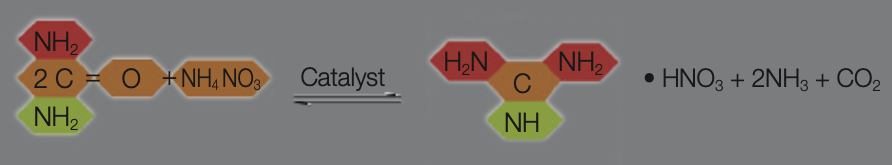
Manufacture of Guanidine Nitrate
Guanidine Nitrate is manufactured from an ammonium nitrate/urea melt in reactors at 180–190 degrees Celsius. This method is the most modern. Of course other production routes can also be used.
The actual raw materials are urea and nitric acid since the ammonium nitrate used is extracted from the neutralization of the produced ammonia and the nitric acid.